The FDA Just Recalled Nearly 1.9 Million Bottles of Water. Here’s What You Need to Know.
The Food and Drug Administration (FDA) has issued a massive bottled water recall.

On May 23, the FDA announced that Natural Waters of Viti Limited is voluntarily recalling over 78,000 cases of Fiji Natural Artesian Water.
Each case contains 24 bottles, which means 1,884,792 individual bottles of water are being recalled.

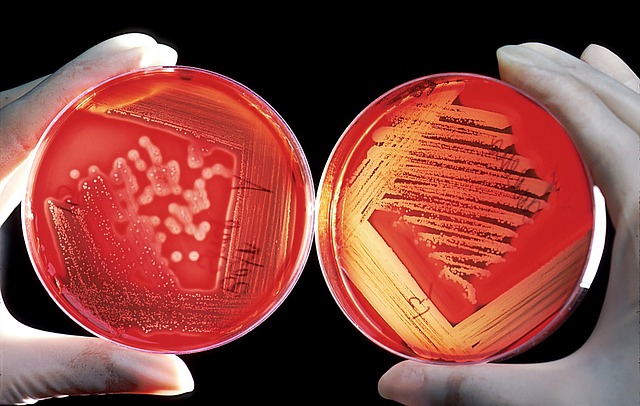
The water bottles have been recalled due to manganese and three bacterial genera, ew.
Manganese is an element that we do need, however, we only need it in trace amounts. Too much manganese can be toxic.

The symptoms of manganese toxicity include tremors, muscle spasms, hearing problems, mania, insomnia, depression, loss of appetite, headaches, irritability, weakness, and mood changes.
National Institutes of Health
That combined with the three bacterial genera is a party my body doesn’t want to entertain.
The recalled water bottles were shipped to Washington state and then made their way throughout America through online sales from Amazon.
If you have Fiji water on hand, you need to check your bottles immediately.

Check the UPC codes and production dates on any bottles that you have.
If you have some bottles included in the recall, contact the retailer where you made your purchase for instructions on returns and refunds.

Here’s the information on the affected cases of Fiji Natural Artesian Water:
- Case UPC Code: 6 32565 00004 3
- Bottle UPC Code: 6 32565 00001 2
- Production Dates: Nov. 11, 2023; Nov. 12, 2023; Nov. 13, 2023; Nov. 24, 2023 and Nov. 25, 2023
For more details and updates on the recall, keep an eye on the FDA’s announcements and the FIJI Water social media posts on X.